Responses of Stomatal Characteristics to Environmental Factors in Fabaceae Trees of the Urban Forest in Maros Regency
Abstract
The response of plants to environmental conditions is reflected in their stomatal characteristics, including stomatal type, number, and density. This novel research examines the influence of environmental factors on the stomatal characteristics of Fabaceae tree leaves in the Urban Forest of Maros Regency. The study explores the relationship between stomatal traits and environmental variables such as light intensity and air humidity. Data analysis employed Pearson correlation to assess how these variables affect stomatal number and density. The results reveal variations in stomatal types among Fabaceae species: Acacia exhibits diacytic stomata, Dadap Merah (Erythrina crista-galli L) has paracytic stomata, Trembesi shows normocytic stomata, and Sengon (Paraserianthes falcataria L) displays cyclocytic stomata. The study also finds that stomatal number and density are higher in the eastern region compared to the western region. Sengon exhibits the highest stomatal number and density among the species studied, whereas Dadap Merah has the lowest. Correlation analysis indicates that only the External Light Intensity of the Stand (ELIS) significantly affects stomatal number and density at the 0.05 (5%) level. Air humidity does not show a significant impact. These findings highlight the role of light intensity in shaping stomatal characteristics in Fabaceae trees within urban forests, contributing to a deeper understanding of plant-environment interactions and their ecological implications.
1. Introduction
The existence of urban forests is a significant factor influencing social and ecological sustainability in cities (Djahangard et al., 2024). Urban forests enhance the well-being of urban residents by providing ecosystem services such as air purification, temperature regulation, and noise reduction (Nowak et al., 2014). Trees naturally absorb carbon dioxide, produce oxygen, and mitigate heat, leading to cooler ambient air temperatures, while also serving as noise reducers (Handayani et al., 2024). Research has demonstrated that urban forests can substantially alleviate urban heat island effects and improve air quality by filtering pollutants (Escobedo et al., 2011). The recommended proportion of green open space in urban areas is at least 30%, according to various urban planning guidelines aimed at ensuring environmental sustainability and public health (WHO, 2016). The area of green open space in Maros Regency ranges from six to seven hectares, with 0.5 hectares designated as an Urban Forest. In the Maros Regency Urban Forest, various types of trees exist, playing an essential role in balancing air quality against activities occurring in urban areas. The diversity of tree species in urban forests bolsters resilience to pests and diseases, thereby ensuring long-term sustainability (Ordóñez & Duinker, 2013).
The function of urban forests in Maros Regency plays an important role in maintaining the balance of the urban ecosystem. And one of the plant families commonly found in urban forests is Fabaceae, which is known for its high adaptability to various environmental conditions (Firmansyah et al., 2022). Stomata on Fabaceae leaves play a key role in the gas exchange process, which affects the efficiency of photosynthesis (Demars et al., 2024). Various environmental factors, such as temperature, humidity, and light intensity, contribute to the formation and distribution of stomata. (Merdekawati, 2015) By understanding the characteristics of stomata on the leaves of Fabaceae trees, we can gain deeper insights into the adaptation of plants to the often-changing urban environment. (Suwandi, 2021) stated that green open spaces can balance CO₂ absorption and O₂ provision. Trees located in urban forest areas greatly affect the air around the urban forest, where trees have the ability to photosynthesize and in the photosynthesis process require stomata and chlorophyll (Juslina, 2022).
The characteristics of stomata were influenced by the environmental conditions where the plants grow (Sinay & Lesilolo, 2020). The number and density of stomata show the response of plants to the environmental conditions where they grow (Merdekawati, 2015). The density and number of stomata were influenced by environmental conditions, where the density of stomata increases as light intensity increases to maximize carbon dioxide absorption (Budiono et al., 2016). One significant environmental factor is light intensity. Increased light intensity stimulates the process of photosynthesis, which in turn increases the plants' need for carbon dioxide (Chauhan et al., 2023). To meet this need, plants adapt by opening more stomata and increasing their density. Research shows that plants exposed to more light tend to have more and broader stomata, which allows for more efficient gas exchange.
Besides light intensity, air humidity also plays a vital role in the regulation of stomata (Cambaba & Kasi, 2022). In high humidity conditions, plants can open their stomata wider to enhance photosynthesis without worrying about losing too much water. However, in low humidity conditions, stomata will close to reduce evaporation, although this can limit photosynthesis capacity. (Raharjo et al., 2015). Therefore, plants must adapt to changing environmental conditions to balance photosynthesis needs and water conservation.
The varying density and number of stomata among plant species indicate different environmental adaptations (Clark et al., 2022). Stomatal density and distribution are influenced by genetic factors as well as environmental conditions such as light intensity, humidity, and CO₂ concentration (Casson & Gray, 2008). In general, plants that grow in drier habitats have a higher stomatal density in response to water stress (Haworth et al., 2023; Sulistiyowati et al., 2021). This adaptation allows plants to optimize water use efficiency and maintain photosynthetic activity under limited water availability (Franks & Farquhar, 2007). Additionally, studies have shown that plants in arid environments often exhibit smaller stomata, which further enhances their ability to regulate water loss (Hetherington & Woodward, 2003). These adaptations highlight the critical role of stomata in plant survival and productivity under varying environmental conditions (Lawson & Blatt, 2014). The research aims to determine the influence and how environmental factors affect the stomatal characteristics of Fabaceae tree leaves in the Urban Forest of Maros Regency.
2. Method
1) Research Area
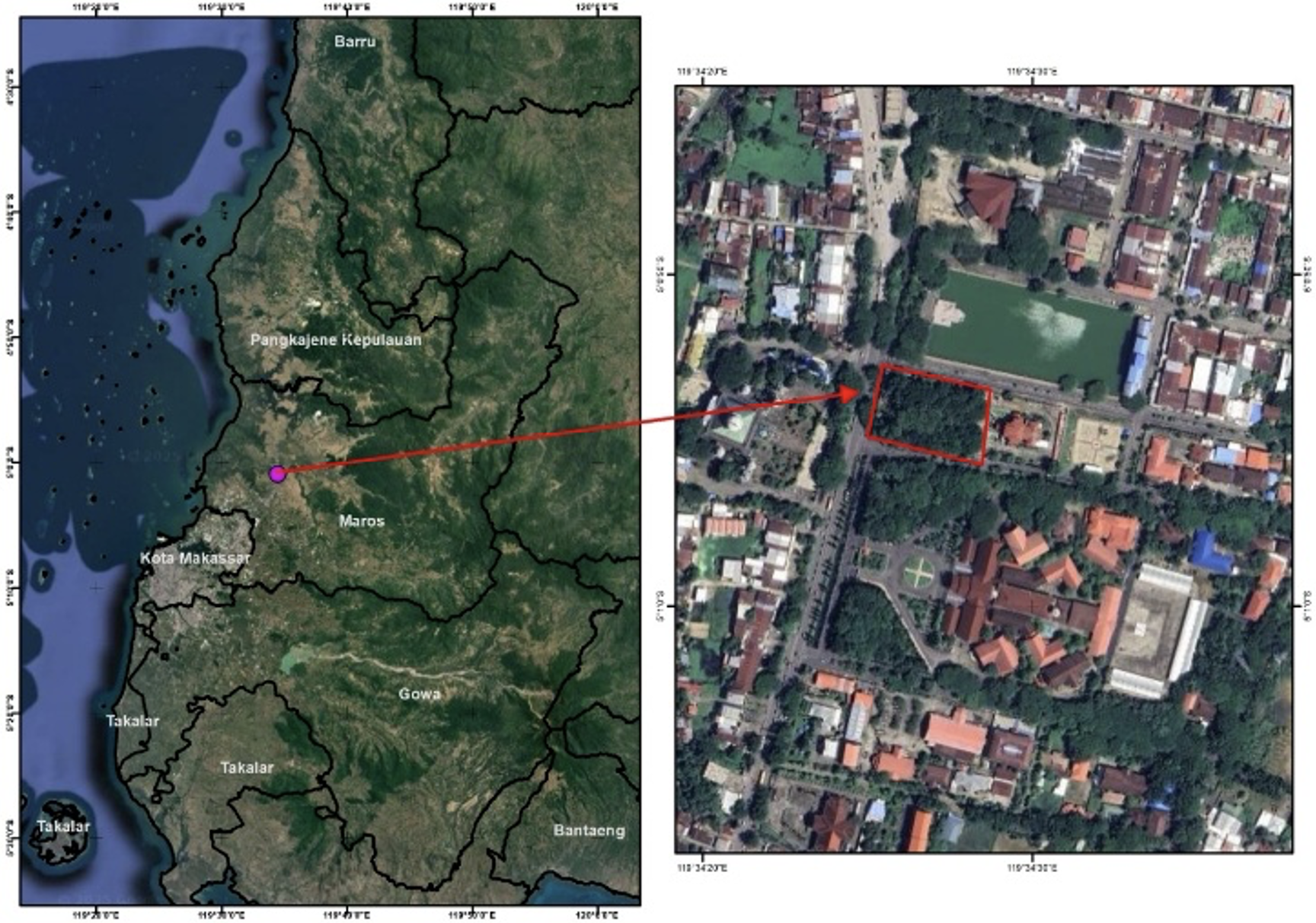
This research was conducted in March-August 2024 in the Urban Forest (Green Open Space) of Maros Regency (figure 1), The study was conducted in the Maros Regency Urban Forest, located in South Sulawesi, Indonesia. The urban forest covers an area of approximately 0.5 hectares and is home to a variety of tree species, including several members of the Fabaceae family. Analysis of research samples were carried out in the laboratory of the Faculty of Agriculture, Hasanuddin University.
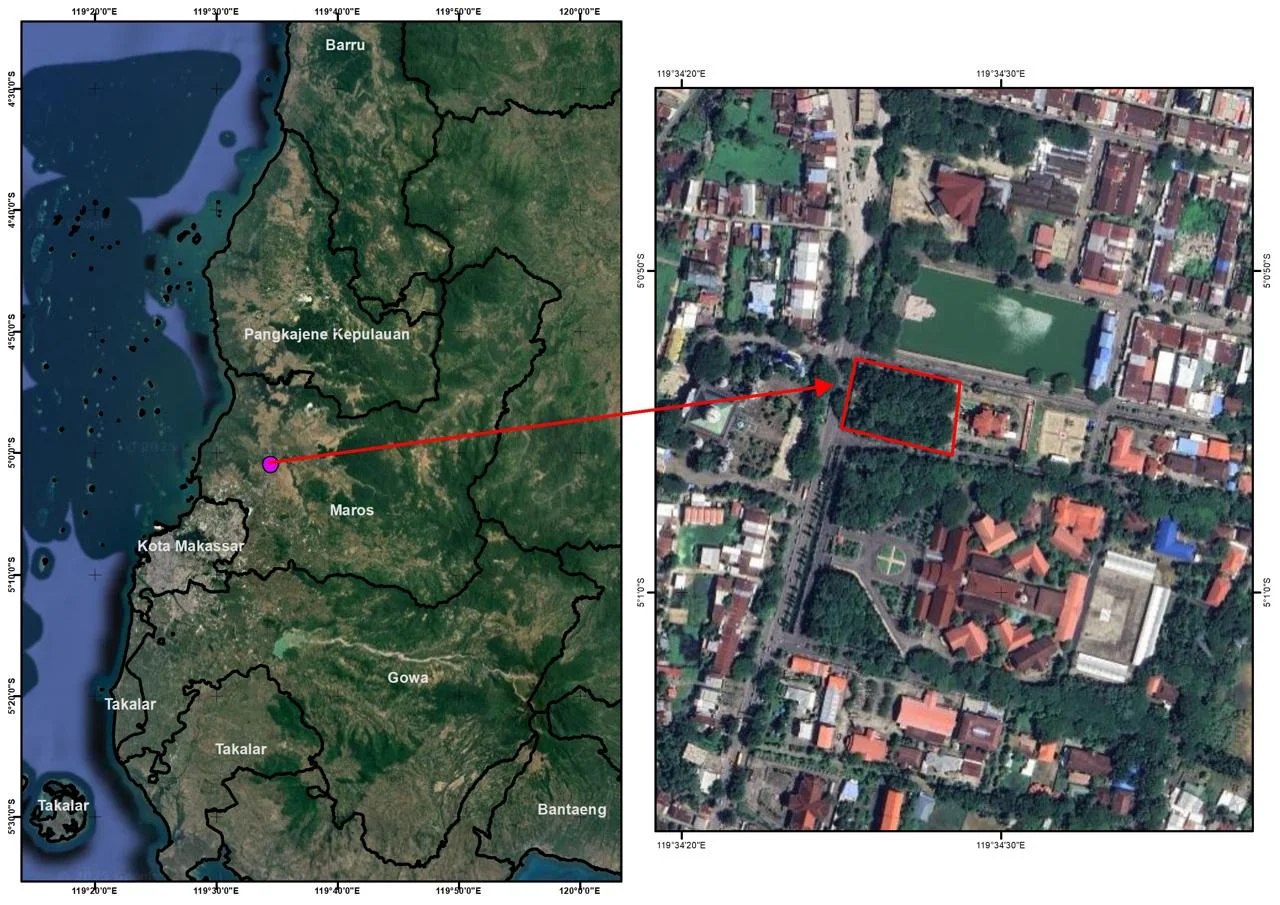
Figure 1. Research location in Maros in the Urban Forest (Green Open Space) of Maros Regency
2) Tools and Materials
The study used tools and materials consisting of Lux Meter, SPAD-502 Plus chlorophyll meter, Binocular Microscope, preparation glass, preparation box (slide box), razor blade (cutter), apparent isolation, pole, digital camera, pen, tally sheet. The materials used in the study consisted of: batteries, clear cuttings, and four types of Fabaceae plants, namely: Acacia tree leaves (Acacia auriculiformis), Dadap Merah plant leaves (Erythirna cristagelli L), Sengon tree leaves (Paraserianthes falcataria L), Trembesi tree leaves (Samanea saman (Jacq.) Merr.)
3) Data Collection Methods and Instruments
The data collected consists of two types. First, primary data is data obtained directly from observations, measurements, calculations, and data analysis directly in the field related to the relationship of stomatal characteristics to environmental factors on the leaves of Fabaceae trees in the Urban Forest of Maros Regency. The second data is secondary data Secondary data is data obtained from literature studies by searching literature through numbers, scientific articles, and books to support research.
4) Sampling and Data Colection
Leaf samples were collected from four Fabaceae species: Acacia auriculiformis, Erythrina cristagalli L., Paraserianthes falcataria L., and Samanea saman (Jacq.) Merr. For each species, five replicates were taken to ensure representative sampling (table 1). Light intensity and air humidity were measured using a Lux Meter and a hygrometer, respectively.
Table 1. Population of Fabaceaetrees in Maros Regency Urban Forest

Fabaceae trees in the Maros Regency urban forest have populations that can be seen in table 2, with a total population of Fabaceae as many as seven populations with acacia and trambesi populations of one tree each, Red Dadap and Sengon each there are two trees, For sampling, one tree each was taken in each type of Fabaceae and repeated sampling five times in each population sample.
5) Laboratory Analysis
The observation of stomatal type, stomatal number, and stomatal density on Fabaceae leaf types was conducted in the laboratory using a microscope with 400x magnification and a field of view area of 0.025 mm². Stomatal counting is done by counting the number of stomata visible in the observed area or a certain number of leaves. For example, count the number of stomata per unit of leaf area, such as mm² or cm². If different types of leaves or leaf parts are used, perform separate counts for each group. To quantify stomatal density, divide the number of stomata counted previously by the leaf area observed. For example, stomatal density can be expressed as the number of stomata per mm² or cm² of leaf. Stomatal density analysis is calculated using the formula below:
Average stomata:
\[ \small \text{Average stomata} = \frac{Sa_1 + Sa_2 + Sa_3 + \dots + Sa_n}{n} \]
Stomata density:
\[ \small K_{a_n} = \frac{\text{average } Sa}{FV} \]
Description:
Fv : Field View (mm2)
Sa1 : Number of stomata in 1st field of view
Sa2 : Number of stomata in 2nd field of view
Sa3 : Number of stomata in 3rd field of view
San : Number of stomata in the field of view to (n)
6) Data Analysis
The data analysis method uses correlation analysis where the correlation analysis used is the correlation of persons (Crocker & Seber, 1980) to get the correlation value of Person (r) using the formula with the following equation (Stein & Corsten, 1991):
The Pearson correlation coefficient formula:
\[ \small r_n = \frac{N \sum XY - \sum X \sum Y} {\sqrt{(N \sum X^2 - (\sum X)^2)(N \sum Y^2 - (\sum Y)^2)}} \]
Description:
RXy : Person Product Moment Correlation Coefficient
x : Value of Independent Variabel
y : The Value of the Independent Variable
N : Number of Samples
Analysis was used with the SmartStatXL v.3.6.5.5 application to observe the effect of light intensity and air humidity on the number of stomata and stomatal density on the leaves of Fabaceae trees.
3. Result
1) Morphological characteristics of stomata on four Fabaceae species in the urban forest of Maros Regency
Fabaceae trees include plant species that are commonly found in the Urban Forest area and also become road protection trees. Based on observations in the research location of the Maros Regency urban forest, there are 4 types of Fabaceae plants consisting of Acacia (Acacia auriculiformis), Red Dadap (Erythrina cristagelli L), Sengon (Paraserianthes falcataria L), and Trembesi (Samanea saman (Jacq).Merr). The results of observations in the microscope to see the type of leaf stomata in four species of Fabaceae (Acacia auriculiformis, Erythirna cristagelli L, Paraserianthes falcataria L, Samanea saman (Jacq.) Merr.), in the Maros Regency Urban Forest can be seen in Figure 2.
In Figure 2, it can be seen that the leaves of Acacia type have diasitic type of stomata, for red dadap Merah type has parasitic type of stomata, trembesi has anomocytic type of stomata, and sengon type has cyclositic type of stomata. Stomata in Acacia auriculiformis (Akasia) species with diasitic type can be seen in (Figure 1.A), where in the stomata there is a closing cell and surrounded by two neighbouring cells with a perpendicular location to the axis of the closing cell. In Figure 1. B stomata on the type of Erythirna cristagelli L (Dadap Merah) with parasitic stomata type, with the characteristics of the stomata, there is one closing cell accompanied by one or more neighboring cells with unequal size. The type of stomata in the Samanea saman (Jacq.) Merr.) (Trembesi) leaf type is anomositik, as shown in Figure 1. C can see the stomata have a cover cell surrounded by cells that resemble epidermal cells both in shape and size. The Paraserianthes falcataria L (Sengon) species with its stomatal type is the cyclomatic type, which has four or more neighboring cells surrounding the stomata in a circular pattern (Figure 1.D). Although the four species observed are plant species from the same family, Fabaceae, they have different types of stomata in each species as obtained from the results of this study; this is because the habitat and water content of the environment or place of growth are different (Prastika et al., 2023). Internal factors, namely the genetics of each species, cause differences in the type and number of stomata in each species. (Latifa et al., 2022).
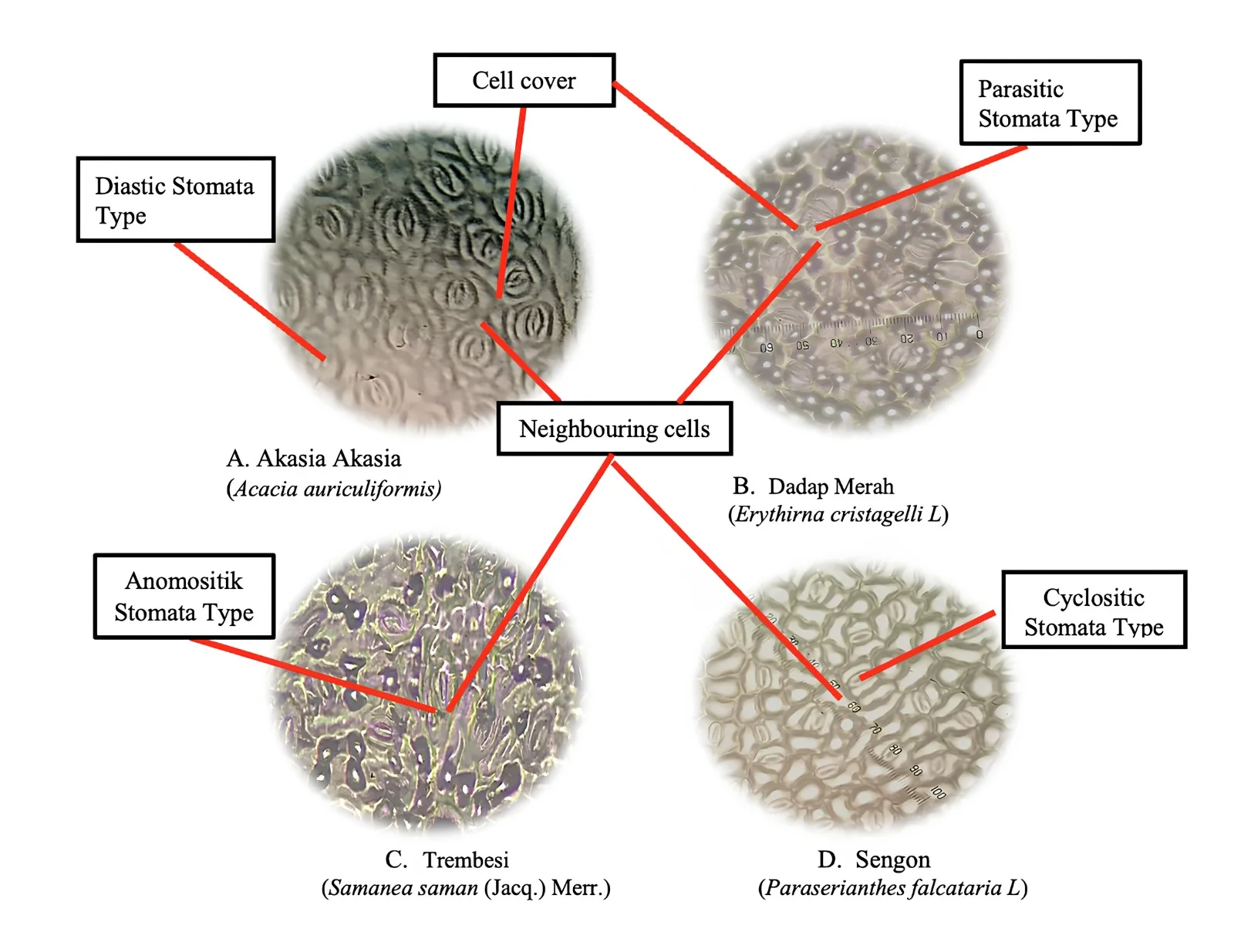
Figure 2. Microscopic Viewing of Fabaceae Tree Stomata with 400 times magnification and a field of view area of 0.025 mm²
2) Number and Density of Stomata in four Fabaceae Tree Species in the Urban Forest of Maros Regency
The results of the analysis of the number and density of stomata in Fabaceae tree species (Acacia auriculiformis, Erythirna cristagelli L, Paraserianthes falcataria L, Samanea saman (Jacq.) Merr.)) in the urban forest of Maros Regency are shown in Table 2.
Table 2. Number and Density of Stomata in four Fabaceae Tree Species in the Urban Forest of Maros Regency

Description: ES= Eastern Stomata, WS= West Stomata
Of the four Fabacaeae tree species shown in table 2, the Sengon species has the highest number of stomata (190,401) and stomatal density (1,259,534.97), while the Dadap Merah species has the lowest number of stomata (810) and stomatal density (5,358.28). Table 1 also shows that the number and density of Western stomata are lower than the number and density of Eastern stomata. The results showed that the higher the number of stomata in plants, the higher the stomatal density. The density and number of stomata were influenced by environmental conditions, where the higher the light intensity, the higher the density (Budiono et al., 2016). In line with the research results (Meriko & Abizar, 2017) which state that the number of stomata affects the density of stomata, namely, if the number of stomata is large, the stomatal density is also higher. However, light intensity that is too high is also not good for plant growth, so stomata need optimum light intensity to maximize stomatal work. In the morning, the light intensity is low, which optimizes the work of the stomata, compared to the afternoon, which has a high light intensity, resulting in excessive water evaporation.
3) Relationship between the number and density of stomata and environmental factors in the leaves of Fabaceae trees in the Maros Regency Urban Forest
Analysis of the relationship between the number and density of stomata to environmental factors on fabaceae tree leaves in the urban forest of maros regency is shown in table 3.
The correlation analysis of light intensity with the number of stomata and stomatal density showed (table 3) that only External Light Intensity of the Stand (ELIS) had a significant effect at the 0.05 (5%) level. In contrast, the Stand Light Intensity (SLI) does not significantly affect the number of stomata and stomatal density. The results of this study indicate that any increase in External Light Intensity of the Stand (ELIS) will have a significant effect at the level of 0.05 (5%) on the ability of the number and density of Eastern stomata to absorb light by (-0.74), meaning that any increase in External Light Intensity of the Stand (ELIS) will reduce the ability of the number and density of Eastern stomata to absorb light intensity by -74%. The measurement results of the air humidity factor, both standing humidity (SH) and External Humidity of the Stand (EHS) in the correlation results, did not significantly affect the number of stomata and stomatal density. The number and density of stomata in each type of Fabaceae tree are different, where the highest number of stomata and stomatal density level is found in the type of session, while the number of stomata and the lowest stomatal density level is in the type of Dadap Merah.
Table 3. Correlation Analysis of Light Intensity and Air Humidity on the Number and Density of Stomata in Fabaceae.

Description: SLI= Stand Light Intensity, ELIS= External Light Intensity of the Stand, SH= Stand Humidity, EHS= External Humidity of the Stand, ES= Eastern Stomata, WS= West Stomata. *) Significant at the real level of 0.05, The number on the body of the table is the value of the correlation coefficient.
The results of this study were supported by research (Carranza-Ramírez et al., 2025), which states that each plant cell has a different sensitivity and will affect the response in plants even given the same light intensity. The resulting research is also supported by research results (Juairiah, 2014), which state that differences in the number of stomata and stomatal density are influenced by internal factors, namely genetic differences, and external factors, namely the growing environment. (Rachmawati & Pujawati, 2020) stated that stomatal density is a genetic trait (heredity). This is supported by the results of research (Meriko & Abizar, 2017), which states that the level of stomatal density in each type of plant is different and influenced by the environment, such as water availability, temperature, CO₂ concentration, and light intensity. One of them is that the higher the light intensity, the higher the density of stomata on the leaf surface will also increase.
4. Discussion
The stomatal characteristics of the four Fabaceae species vary in terms of stomatal type, which differs among each species. This variation is influenced by environmental and genetic factors. Environmental factors affecting stomatal type include light intensity and air humidity. Although all six plant species belong to the same family, they exhibit different stomatal types due to differences in their habitats and water availability in their growing environments (Prastika et al., 2023). Internal factors, such as the genetic makeup of each species, also play a significant role in determining the type and number of stomata (Latifa et al., 2022).
The stomatal type of the Acacia leaf is diacytic, characterized by two subsidiary cells surrounding the guard cells, positioned perpendicular to the long axis of the guard cells. Banyan and Capilong plants exhibit paracytic stomata, while palm has mesoperigenous stomata, and Acacia has diacytic stomata (Papuangan et al., 2014). The Red Dadap (Erythrina) species has paracytic stomata, where the long axis of the guard cells is parallel to the subsidiary cells, while the Trembesi (Rain Tree) species has anomocytic stomata, where the number of subsidiary cells surrounding the guard cells is indeterminate and indistinguishable from other epidermal cells.
Three types of stomata were observed in this study: the Rain Tree (Ki Hujan) leaf has anomocytic stomata, the Butterfly Tree (Kupu-kupu) leaf has anisocytic stomata, and the Red Dadap (Dadap Merah) has paracytic stomata (Jaya et al., 2014). The Sengon (Albizia) species exhibits cyclocytic stomata, where four or more subsidiary cells form a ring-like structure around the stomata. Cyclocytic stomata are characterized by four or more subsidiary cells arranged in a circular pattern surrounding the stomata (Evert, 2006).
The results of the study on the number and density of stomata in Fabaceae plants indicate that the number and density of stomata on the western side are lower compared to those on the eastern side. The higher the light intensity received by the trees, the greater the number and density of stomata. Stomatal density and number are influenced by environmental conditions, where higher light intensity leads to higher stomatal density (Budiono et al., 2016). Additionally, this is supported by research from (Meriko & Abizar, 2017), which states that the number of stomata affects stomatal density, meaning that a higher number of stomata results in greater stomatal density. However, excessively high light intensity is also not beneficial for plant growth, as stomata require optimal light intensity to function efficiently. In the morning, light intensity is not too high, allowing stomata to work more optimally compared to midday, when light intensity is high, leading to excessive water evaporation. Genetic factors and the differing environmental conditions of the plant's habitat also influence stomatal characteristics. Furthermore, light intensity, water availability, temperature, and CO₂ concentration are additional factors that affect the number and density of stomata (Juma’ani & Munawwaroh, 2017).
The number and density of stomata vary among each Fabaceae tree species, with the highest stomatal count and density found in the Sengon (Albizia) species, while the lowest stomatal count and density are observed in the Dadap Merah (Erythrina) species. These findings are supported by research from (Salisbury & Ross, 1995), which states that each cell in plants has varying sensitivities, influencing the plant's response even when exposed to the same light intensity. The results are further corroborated by a study from (Juairiah, 2014), which explains that internal factors, such as genetic variation, and external factors, such as the environmental conditions of the growing site influence differences in stomatal nu. Rachmawati and Pujawati (2020) state that stomatal density is a genetic trait (inherited). This is supported by research from (Meriko & Abizar, 2017), which indicates that stomatal density varies among plant species and is influenced by environmental factors such as water availability, temperature, CO₂ concentration, and light intensity. For instance, higher light intensity leads to an increase in stomatal density on the leaf surface.
The relationship between stomatal number, stomatal density, and environmental factors—specifically light intensity and air humidity—shows that only External Light Intensity has a significant influence. Each increase in External Light Intensity affects the ability of the stomata on the eastern and western sides to absorb light. As External Light Intensity increases, the stomata tend to close to prevent excessive water evaporation, thereby reducing light absorption by the stomata. This is supported by research from Sumadji (2020), which states that stomatal closure is an adaptation mechanism to drought stress, reducing transpiration rates and preventing water loss during periods of high light intensity and temperature. Additionally, research by Sudomo (2009) indicates that both extremely low and extremely high light intensities negatively impact plant growth by slowing it down, as they affect the functioning of stomatal cells in the leaves. Each tree species has an optimal range of light intensity, within which stomatal function can operate efficiently. Sudomo (2009) further emphasizes that optimal light intensity is crucial for maximizing plant growth. However, Light Intensity, Stand Humidity, and External Humidity do not have a significant effect on stomatal number and density. Future research should explore the effects of varying CO₂ concentrations and soil moisture on stomatal behavior, as these factors were not addressed in this study. Additionally, long-term monitoring of stomatal adaptations to seasonal changes and extreme climate conditions could provide deeper insights into plant resilience. Genetic analysis of stomatal traits and comparative studies across different ecosystems would further enhance our understanding of stomatal adaptations in Fabaceae species
5. Conclusion
This study analysed the relationship of stomatal characteristics in Fabaceae tree leaves with environmental factors, specifically light intensity and air humidity in the Maros City Forest. The results showed that the type, number and density of stomata varied among Fabaceae species, reflecting adaptation to specific environmental conditions. External light intensity (ELIS) had a significant effect on stomatal number and density with a negative correlation at the 5% significance level. In contrast, light intensity within the stand (SLI) had no significant effect because the trees provided shading that stabilised the light intensity. Air humidity inside and outside the stand had no significant impact on stomata as the variation was small in the urban forest environment. Other factors, such as plant genetics and carbon dioxide concentration, are more dominant in influencing stomatal characteristics.
This study has limitations, such as not considering seasonal variability as well as other factors such as carbon dioxide concentration, soil quality, and air temperature. Addressing these variables in future research could provide a more comprehensive understanding of the environmental drivers influencing stomatal behaviour. Further research is recommended to explore the genetic mechanisms underlying stomatal differentiation and to expand the study to other plant families within the Maros Regency urban forest. Such investigations would enhance our knowledge of plant adaptive strategies and support the development of effective conservation practices. Moreover, integrating physiological measurements, such as transpiration rates and photosynthetic efficiency, could provide a more holistic understanding of how stomatal behaviour influences plant ecology.
6. Author Contributions
The first author (RY) contributes as a compiler and data analyzer based on the research results. In contrast, the second author (HS) contributes methods to help interpret data and research results, and the third (N) and fourth authors (IND) contribute to help prepare the article.
7. Competing Interests
All authors declare no conflicts of interest regarding this publication.
8. Acknowledgements
Thanks to the Indonesian Research and Technology Agency (BRIN) for providing funding for thesis research through the BARISTA program, the Hasanuddin University Faculty of Agriculture Integrated Laboratory for its facility support, and to the Supervisors from BRIN and the Forestry Study Program, Faculty of Agriculture and Forestry, Maros Muslim University who have spent much time for the preparation of this research
9. References
Cambaba, S. & Kasi, P. D. (2022). Karakteristik Stomata Daun Pucuk Merah (Syzygium oleana) Berdasarkan Waktu Pengambilan Sampl Yang Berbeda. Cokroaminoto Journal of Biological Science, 4(1), 19-25. Retrieved from https://science.e-journal.my.id/cjbs/article/view/95
Carranza-Ramírez, J. E., Borda, A. M. & Moreno-Fonseca, L. P. (2025). LED light modifies plant architecture, physiological parameters and cannabinoid content in three varieties of Cannabis sativa L. South African Journal of Botany, 176, 231–240. https://doi.org/10.1016/j.sajb.2024.11.023
Casson S, Gray JE. Influence of environmental factors on stomatal development. New Phytol. 2008;178(1):9-23. https://doi.org/10.1111/j.1469-8137.2007.02351.x
Chauhan, J., Prathibha, M. D., Singh, P., Choyal, P., Mishra, U. N., Saha, D., Kumar, R., Anuragi, H., Pandey, S., Bose, B., Mehta, B., Dey, P., Dwivedi, K. K., Gupta, N. K. & Singhal, R. K. (2023). Plant photosynthesis under abiotic stresses: Damages, adaptive, and signaling mechanisms. In Plant Stress (Vol. 10). Elsevier B.V. https://doi.org/10.1016/j.stress.2023.100296
Clark, J. W., Harris, B. J., Hetherington, A. J., Hurtado-Castano, N., Brench, R. A., Casson, S., Williams, T. A., Gray, J. E. & Hetherington, A. M. (2022). The origin and evolution of stomata. In Current Biology (Vol. 32, Issue 11, pp. R539–R553). Cell Press. https://doi.org/10.1016/j.cub.2022.04.040
Crocker, D. C. & Seber, G. A. F. (1980). Linear Regression Analysis. In Technometrics (Vol. 22, Issue 1). https://doi.org/10.2307/1268395
Demars, B. O. L., Schneider, S. C., Thiemer, K., Dörsch, P., Pulg, U., Stranzl, S., Velle, G. & Pathak, D. (2024). Light and temperature controls of aquatic plant photosynthesis downstream of a hydropower plant and the effect of plant removal. Science of the Total Environment, 912. https://doi.org/10.1016/j.scitotenv.2023.169201
Djahangard, M., Buckwitz, S., Hioki, C., He, J., Zhou, Z., Zhang, H. & Yousefpour, R. (2024). A global comparative analysis of local recreation behaviors and values in peri-urban forests. Environmental and Sustainability Indicators, 24. https://doi.org/10.1016/j.indic.2024.100525
Escobedo, F. J., Kroeger, T., & Wagner, J. E. (2011). Urban forests and pollution mitigation: Analyzing ecosystem services and disservices. Environmental Pollution, 159(8-9), 2078-2087. https://doi.org/10.1016/j.envpol.2011.01.010
Franks PJ, Farquhar GD. The mechanical diversity of stomata and its significance in gas-exchange control. Plant Physiol. 2007 Jan;143(1):78-87. https://doi.org/10.1104/pp.106.089367
Haworth, M., Marino, G., Atzori, G., Fabbri, A., Daccache, A., Killi, D., Carli, A., Montesano, V., Conte, A., Balestrini, R., & Centritto, M. (2023). Plant Physiological Analysis to Overcome Limitations to Plant Phenotyping. Plants, 12(23), 4015. https://doi.org/10.3390/plants12234015
Lawson T, Blatt MR. Stomatal size, speed, and responsiveness impact on photosynthesis and water use efficiency. Plant Physiol. 2014 Apr;164(4):1556-70. https://doi.org/10.1104/pp.114.237107.
Meriko, L. & Abizar. (2017). Struktur Stomata Daun Beberapa Tumbuhan Kantong Semar (Nepenthes spp.) [Structure of Leaves Stomata on Some Pitcher Plants (Nepenthes spp.). Jurnal Ilmu-Ilmu Hayati, 16(3), 325–330. https://doi.org/10.14203/beritabiologi.v16i3.2398
Nowak, D. J., Hirabayashi, S., Bodine, A., & Greenfield, E. (2014). Tree and forest effects on air quality and human health in the United States. Environmental Pollution, 193, 119-129. https://doi.org/10.1016/j.envpol.2014.05.028
Ordóñez, C., & Duinker, P. N. (2013). An analysis of urban forest management plans in Canada: Implications for urban forest management. Landscape and Urban Planning, 116, 36-47. https://doi.org/10.1016/j.landurbplan.2013.04.007
Papuangan. N., Nurhasanah., M. D. (2014). Jumlah Dan Distribusi Stomata Pada Tanaman Penghijauan Di Kota Ternate. 3(September). https://doi.org/https://doi.org/10.33387/bioedu.v2i1.62
Prastika, D., Sarjani, T. M., Mahyuni, S. R., Hariani, I., Ramadhan, D. A., Rezeki, S., Tiara, R., Hendrik, E., Aulia, R. & Amalia, T. (2023). Identifikasi Tipe Stomata Anggota Suku Myrtaceae di Kota Langsa. Jurnal Sains Dan Edukasi Sains, 6(1), 20–27. https://doi.org/10.24246/juses.v6i1p20-27
Rachmawati, R. N. & Pujawati, E. D. (2020). Karakteristik Stomata Nyawai (Ficus variegata Blume) dari 3 Sumber Benih Aasal Kalimantan di KHDTK Riam Kiwa Desa Lobang Baru The Characteristics of Stomata Nyawai (Ficus variegata Blume) From 3 Sources of Origin Seeds Kalimantan in KHDTK Riam Kiwa Lobang. Jurnal Sylva Scienteae, 3(6), 1078–1085. https://doi.org/10.20527/jss.v3i6.4725
Raharjo, H. P., Haryanti, S., & Hastuti, R. B. (2015). Pengaruh Tingkat Kepadatan Lalu Lintas dan Waktu Pengamatan yang Berbeda Terhadap Ukuran dan Jumlah Stomata Daun Glodokan (Polyalthia longifolia. Sonn). Jurnal Akademika Biologi, 4(1), 73-84. Retrieved from https://ejournal3.undip.ac.id/index.php/biologi/article/view/19402
Sinay, H. & Lesilolo, M. H. (2020). Karakterisasi Stomata Daun Pegagan (Centella asiatica (L.) Urban.) pada Ketinggian Wilayah Berbeda di Pulau Ambon. Science Map Journal, 2(1), 1–7. https://doi.org/10.30598/jmsvol2issue1pp1-7
Stein, A. & Corsten, L. C. A. (1991). Universal Kriging and Cokriging as a Regression Procedure. Biometrics, 47(2), 575. https://doi.org/10.2307/2532147
Sudomo, A. (2009). Pengaruh naungan terhadap pertumbuhan dan mutu bibit manglid (Manglieta glauca BI). Tekno hutan tanaman, 2(2), 59-66.
Sumadji, Angga Rahabistara. (2020). “Kerapatan Stomata Dan Kaitannya Terhadap Kekeringan Pada Tanaman Padi Varietas IR64.” Widya Warta Jurnal Ilmiah Tahun XLIV(1): 43–54. http://portal.widyamandala.ac.id/jurnal/index.php/warta/article/view/907
Suwandi, R. (2021). Fungsi dan Manfaat Ruang Terbuka Hijau Di Kelurahan Kampung Bandar Kecamatan Senapelan Kota Pekanbaru. Universitas Islam Riau. http://repository.uir.ac.id/id/eprint/18805
Copyright (c) 2025 Jurnal Wasian

This work is licensed under a Creative Commons Attribution 4.0 International License.
Copyright and License
All articles published in Wasian Journal are the property of the authors. By submitting an article to Wasian Journal, authors agree to the following terms:
-
Copyright Ownership: The author(s) retain copyright and full publishing rights without restrictions. Authors grant the journal the right to publish the work first and to distribute it as open access under a Creative Commons Attribution 4.0 International License (CC BY 4.0).
-
Licensing: Articles published in Wasian Journal are licensed under a Creative Commons Attribution 4.0 International License (CC BY 4.0). This license allows others to share, copy, and redistribute the material in any medium or format, and adapt, remix, transform, and build upon the material for any purpose, even commercially, provided that proper credit is given to the original author(s) and the source of the material

This work is licensed under a Creative Commons Attribution 4.0 International License. -
Author's Rights: Authors are permitted and encouraged to post their work online (e.g., in institutional repositories or on their website) prior to and during the submission process, as it can lead to productive exchanges and greater citation of published work.
-
Third-Party Content: If your article contains material (e.g., images, tables, or figures) for which you do not hold copyright, you must obtain permission from the copyright holder to use the material in your article. This permission must include the right for you to grant the journal the rights described above.
-
Reprints and Distribution: Authors have the right to distribute the final published version of their work (e.g., post it to an institutional repository or publish it in a book), provided that the original publication in Wasian Journal is acknowledged.
For the reader you are free to:
- Share — copy and redistribute the material in any medium or format for any purpose, even commercially.
- Adapt — remix, transform, and build upon the material for any purpose, even commercially.
- The licensor cannot revoke these freedoms as long as you follow the license terms.
Under the following terms:
- Attribution — You must give appropriate credit , provide a link to the license, and indicate if changes were made . You may do so in any reasonable manner, but not in any way that suggests the licensor endorses you or your use.
- No additional restrictions — You may not apply legal terms or technological measures that legally restrict others from doing anything the license permits.
Notices:
You do not have to comply with the license for elements of the material in the public domain or where your use is permitted by an applicable exception or limitation .
No warranties are given. The license may not give you all of the permissions necessary for your intended use. For example, other rights such as publicity, privacy, or moral rightsmay limit how you use the material.